Get Certified in Radiation Oncology (RO)
Learn the steps to becoming ABR board certified in radiation oncology, including eligibility requirements and the exam process. Visit our alternate pathways page to explore prerequisites, eligibility requirements, and other details for the radiation oncology pathway.
-
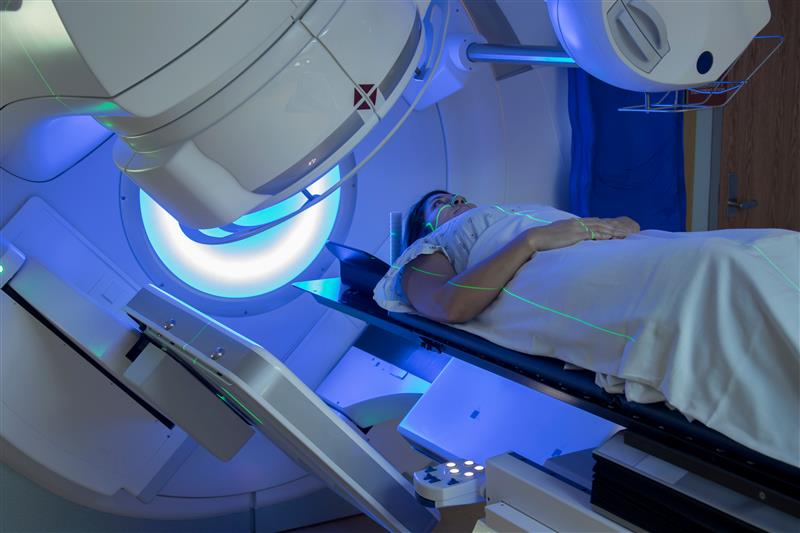
Radiation oncologists use ionizing radiation to treat cancer and other conditions.
The RO certification process ensures you have the knowledge and skills to provide high-quality patient care. You’ll need to meet specific education, training, and exam requirements to be eligible for certification.
Eligibility Requirements
- Complete a year of clinical training
Before starting residency, you must complete a year of clinical training in the U.S. or Canada, accredited by the Accreditation Council for Graduate Medical Education (ACGME), Royal College of Physicians and Surgeons of Canada (RCPSC), or American Osteopathic Association (AOA).
Accepted disciplines:
- Internal medicine
- Pediatrics
- Surgery (or surgical specialties)
- Obstetrics and gynecology
- Family practice
- Transitional or categorical radiation oncology
Specialties other than those listed, including pathology and psychiatry, don’t count toward this requirement.
- Complete an accredited residency program
Complete a four-year ACGME- or RCPSC-accredited RO residency. It must:
- Include 36 months of clinical radiation oncology with exposure to pathology and medical oncology.
- Include no more than six months in elective rotations outside the parent institution, as defined in the RRC’s program requirements.
Interested in research? Check out the Holman Research Pathway for an alternate option.
- Pass your exams
- Qualifying Exam – Includes three portions:
- Medical Physics for Radiation Oncology (taken after 32 months of radiation oncology training, or after 24 months if approved in writing by your program director)
- Radiation and Cancer Biology (taken after 32 months of radiation oncology training, or after 24 months if approved in writing by your program director)
NOTE: Your program must contact the ABR at information@theabr.org to request an exception for you to take the Medical Physics for Radiation Oncology and/or the Radiation and Cancer Biology exam(s) early. The process requires a form signed by you and your program director.
- Clinical Radiation Oncology (taken after 44 months of radiation oncology training)
- Oral Certifying Exam – You can take the Oral Certifying Exam the calendar year following graduation from your residency and after passing the Qualifying Exam.
- Hold a valid state or provincial medical license
If you’re still in training, a valid training license is acceptable.
- Demonstrate ethical standards
The ABR expects all candidates to uphold fundamental moral and ethical principles and exhibit professional behavior throughout the certification process.
Board Eligibility
Board eligibility is the time from when you finish your training to when you receive initial board certification. You have six full calendar years from residency completion to complete your RO certification.
To learn more, read our Board Eligibility Policy.
Residency Requirements and Policies
Residency Completion
Effective July 1, 2026, you must complete your four-year residency within eight years.
Taking Leave
Personal and family needs are important during residency. We support you in taking leave when you need it. To learn more, read our Residency Completion Policy.
To learn more, read our Residency Leave Policy.
-
Apply for Certification
Once you begin your residency, your program director or coordinator will submit your name and email address to the ABR, so we’ll know you have officially started training.
As soon as we receive your information, we’ll send a welcome notification with instructions on how to log in to myABR and start your application. You’ll need:
- Medical school information
- Internship certificate (PDF copy)
- Residency start date
- Residency training dates by year
- ABNM certification, if applicable
You can submit your application annually between July 1 and October 31. The application fee is due when you apply. Applications are reviewed in the order they’re received.
-
Exam Process
Certification exams assess your knowledge, skills, and readiness for independent practice. The RO certification process includes two exams:
- Qualifying
- Oral Certifying
Qualifying Exam
The Qualifying Exam is a computer-based exam that has three parts:
- Medical Physics for Radiation Oncology
- Radiation and Cancer Biology
- Clinical Radiation Oncology
Medical Physics for Radiation Oncology
Exam Content
This exam tests foundational knowledge of medical physics as applied to radiation oncology. Topics include:
- Basic physics
- Radiation measurements
- Treatment planning
- Imaging modalities and applications in radiotherapy
- Brachytherapy
- Advanced treatment planning and special procedures
- Safety, QA, and radiation protection
Format
This computer-based exam takes approximately two and a half hours, delivered in one session. Explore exam formats in more depth in the Qualifying Exam Guide, including a breakdown of how many questions are in each section.
Timing
The ABR offers this exam once a year (typically in June).
Radiation and Cancer Biology
Exam Content
This exam tests your knowledge of the principles of radiation and cancer biology underlying the practice of radiation oncology, including:
- Interaction of radiation with matter
- Molecular and cellular damage and repair
- Cellular responses to radiation
- Linear energy transfer (LET) and oxygen effect
- Tumor biology and microenvironment
- Cancer biology
- Radiobiology of normal tissues
- Dose delivery
- Combined modality therapy
- Late effects and radiation protection
Format
This computer-based exam takes approximately two and a half hours, delivered in one session. Explore exam formats in more depth in the Qualifying Exam Guide, including a breakdown of how many questions are in each section.
Timing
The ABR offers this exam once a year (typically in June).
Clinical Radiation Oncology
Exam Content
This exam assesses your understanding of the entire field of oncology and radiation oncology. For details on the domains that are included, please see the exam content study guides.
Format
This computer-based exam takes approximately six hours, delivered across two sessions on one day. Explore exam formats in more depth in the Qualifying Exam Guide, including a breakdown of how many questions are in each section.
Timing
The ABR offers this exam once a year (typically in May).
Qualifying Exam Scoring
The Qualifying Exam parts are scored using a criterion-referenced scoring method. Your score is based on how well you do compared with a fixed standard — not how everyone else does.
Each part of the Qualifying Exam is scored as a whole, and the result is either pass or fail.
- All categories or modules on each part must be passed together.
- If you don’t meet the standard, you must retake that entire part of the exam (i.e. physics, radiobiology, or clinical).
- You must pass each part to pass overall. You can retake them separately if you only fail one.
Learn more about exam scoring.
Oral Certifying Exam
Exam Content
The Certifying Exam is an oral, case-based exam where you’ll discuss real clinical scenarios. You’ll answer questions on a range of topics, including:
- Anatomy
- Epidemiology
- Pathology
- Patterns of local, regional, and distant involvement
- Clinical evaluation, including staging and tumor markers
- Selection and integration of treatment modalities, including surgery and systemic therapy
Cases are curated to sample from the various large clinical domains
Format
Eight 25-minute oral exams covering the eight clinical categories are conducted one-on-one each with different examiners. Explore exam formats in more depth in the Oral Certifying Exam Guide.
Timing
The ABR offers this exam twice a year (typically in April and October).
Scoring
The Oral Certifying Exam is scored using a criterion-referenced scoring method. Your score is based on how well you do compared with a fixed standard — not how everyone else does.
The Oral Certifying Exam is scored by content category, and the result is pass, conditional pass, or fail.
- If you fail one or two categories, you’ll receive a conditional pass and you’ll only need to retake the portions you did not pass — not the entire exam. When you retake the exam, two examiners will re-evaluate you in each conditioned category. If both pass you, the condition will be removed.
- If you fail three or more categories, you’ll need to retake the entire exam.
Learn more about exam scoring.
-
Exam Registration
Once you’re eligible to take an exam, the ABR will send you an email invitation three months before your Qualifying Exam, and five months before your Oral Certifying Exam. The email will include instructions on completing registration in myABR.
To ensure you receive these emails, update your contact information, add @theabr.org to your approved senders list, and keep an eye on your spam folder.
Before registering for an exam, you’ll need to pay all exam fees.
-
Exam Preparation
Experienced volunteers from all different practice settings create ABR exams. We offer study guides and exam blueprints to help you prepare, but you’ll need to find the study materials that work for you. Keep in mind materials from other organizations won’t always match the format, scope, or difficulty of ABR exams.
To learn more about how exams are created and scored, please see:
- Information for volunteers – how volunteers create exam questions
- Illustrated life cycle of an ABR exam item– a look at the extensive QA process that each question goes through
- Exam scoring and results – how we evaluate your exam
Studying for RO Exams
In radiation oncology exams, disease staging questions follow the latest American Joint Commission on Cancer guidelines (currently 8th edition, 2016). However, if a question is based on an older clinical trial, staging will match what was used in that study.
For toxicity classification, the exams use the most recent Common Terminology Criteria for Adverse Events version (currently 5.0, 2017). Again, if the question is about a past clinical trial, it’ll refer to the system that was in place at the time of the trial.
Study Guides and Sample Questions
- Medical Physics for Radiation Oncology Study Guide
- Radiation and Cancer Biology Study Guide
- Clinical (General and Radiation Oncology) Study Guide
- Oral Exam Study Guide + Reenactment Video
- RO Non-Clinical Skills Syllabus
- Qualifying Exam Guide
- Oral Certifying Exam Guide
Exam Readiness Checks
After you’ve registered for an upcoming computer-based exam, you’ll receive a link to the Exam Readiness Check. You can also access it by logging in to myABR.
This quick check helps you get comfortable with the exam interface, navigation, and tools before exam day.
Here’s how it works:
- Do the readiness check exactly like you’ll take the exam – on the same computer and in the same location.
- Once you’ve launched the readiness check, a quick technical verification will check your camera, microphone, internet, and software. Learn more about technical requirements for remote exams.
- You’ll be required to complete an identification check by scanning your ID and taking a selfie.
If you change computers or locations before exam day, we recommend repeating the check to ensure everything works as expected.
Sample Questions
Once you complete the Exam Readiness Check, you’ll get access to a set of sample questions. They’ll help you get familiar with the different question formats you can expect on the exam, such as:
- Multiple-choice – Most questions follow this format
- Drag and drop – You’ll have to identify an anatomical site by clicking on the correct spot in an image.
While taking your exam, you can flag items within sections to go back to if you have time. Once you leave a section, you cannot return to it.
Answer Sheets for Sample Questions
Using Calculators in Exams
You may need to use a calculator for the RO Qualifying Exam. The ABR provides an embedded calculator within the exam interface. No personal electronic, computing, or calculator devices are permitted in the exam area, including any devices capable of sending or receiving electronic messages.
Practicing with the Embedded Calculator:
To familiarize yourself with the embedded calculator:
- Use the practice exam link in myABR after registering.
- If you’re not registered yet, download the Chrome Scientific Calculator extension.
Practice Exercises (for familiarity, not actual exam questions):
Try these to get comfortable with calculator functions:
- Logarithmic functions: Calculate the decay constant of 99mTc from its half-life of 6.02 hr.
- Trigonometric functions: Find the first spherical Bessel function (sin(x)/x) at 0.25 radians.
- Hyperbolic functions: Solve for y in the equation for the Gateway Arch:
y = 693.9 – 68.8 cosh(0.01x)
when x = 100 ft.
- Exponential decay: Determine the remaining activity of an 86-MBq 99mTc source after 14.6 hours.
- Memory function & Bateman equations: Calculate the 99mTc activity in a generator 4 hours after elution, given 99Mo decay parameters.
-
Current Scoring Status
Exam results are available in myABR about one month after an exam. You’ll receive an email notification as soon as your results are posted.
Results for the Radiation Oncology Clinical Qualifying Exam are in the following phase of the scoring process. This graphic will change as the process progresses. Check back regularly after your exam for real-time updates.

Learn more about ABR exam scoring and results.
-
History of Aggregate Exam Results
To help you understand the typical performance of first-time takers on RO exams, we’ve compiled tables showing pass rates from recent qualifying and certifying exams.
Qualifying Exam Results
First-Time Takers (Residents ONLY)
Clinical
Year Percentage Passed Total First-Time Takers 2016 96 200 2017 84 197 2018 96 193 2019 99 150 20201 98 292 2021 99 101 2022 93 204 2023 97 215 2024 95 212 2025 97 186 1Results include December 2020 and April 2021 administrations.
Physics
Year Percentage Passed Total First-Time Takers 2016 97 192 2017 88 203 2018 71 206 2019 98 198 20201 98 297 2021 92 150 2022 98 196 2023 88 205 2024 88 203 2025 86 187 1Results include December 2020 and April 2021 administrations.
Biology
Year Percentage Passed Total First-Time Takers 2016 9 192 2017 89 203 2018 74 206 2019 99 211 20201 99 295 2021 97 149 2022 95 203 2023 93 209 2024 100 203 2025 95 176 1Results include December 2020 and April 2021 administrations.
Certifying Oral Exam Results
First-Time Takers
Year Total First-Time Takers % Passed Complete % Passed Conditional % Failed 2016 207 87 9 4 2017 185 92 5 2 2018 162 92 7 1 2019 209 88 9 3 2021 (April) 184 87 6 7 2021 (August) 184 91 5 3 2022 195 91 7 2 2023 177 86 11 2 2024 193 85 11 3 2025 176 77 18 6 2026 161 88 9 3